The 3rd International Conference on Clinical Trials 2026
Feb 4-5, 2026 | Narcissus The Royal Hotel, Riyadh.
Voice of Hope
Register Now3+
Years Running
250+
Sessions
Attendees
1,000+ Visitors
Speakers
30+ Experts
Voice of Hope
The Third International Conference on Clinical Trials, held on February 4–5, 2026 under the theme “Voice of Hope”, embodies a dynamic global platform where minds converge and expertise intersects, serving as a starting point toward more humane and innovative horizons.
The conference is organized by the King Abdullah International Medical Research Center (KAIMRC), where the Center conducts research and development activities spanning a broad spectrum, from early basic research through translational research to clinical studies, including Phase I clinical trials.
The conference attracts a distinguished group of scientific leaders, decisionmakers, researchers, regulatory bodies, and representatives from both the public and private sectors, to contribute to global collaboration, knowledge exchange, and the sharing of pioneering experiences. It also aims to build effective professional bridges among researchers, sponsors, solution providers, and regulatory authorities, shaping the future of clinical trials through discussions of the latest global trends and established best practices, in full alignment with Saudi Vision 2030, based on the belief that investment in research and innovation is an investment in people, toward building a sustainable and healthy future.
Why This Conference?
The ICCT provides a unique platform for collaboration, innovation, and knowledge sharing.
- Provide an interactive scientific environment
- Enhance collaboration and institutional partnerships
- Enable participants to discuss current challenges
- Anticipate future solutions
- Support the development of clinical trials
- Achieve sustainable impact on community health locally and internationally
Strategic Themes
Key focus areas that will shape the future of technology and clinical research
Building Capacity
Developing infrastructure and capabilities for conducting high-quality clinical trials
AI & Technology
Employing modern technologies and artificial intelligence to advance research
Patient-Centered Approach
Enhancing focus on patient needs, experiences, and outcomes
Ethical Standards
Maintaining high ethical and regulatory standards in research
Sustainable Funding
Developing sustainable funding models for long-term research success
Investigator Leadership
Strengthening the role and leadership of principal investigators
Vision & Mission
Our guiding principles for transforming clinical research
Our Vision
To cultivate a trusted, patient empowered clinical trial ecosystem where patient voices drive innovation and improve health outcomes.
Our Mission
- Center patients as partners in clinical research by elevating their experiences and expectations
- Build trust through ethical, transparent, and culturally sensitive trial practices
- Foster national and global collaboration to improve access and impact
- Advance innovation through AI, decentralized trials, and digital health solutions
- Strengthen a sustainable research ecosystem grounded in quality and continuous improvement
Target Audience
Professionals who will benefit from attending the FutureTech Summit
Conference Objectives
Key goals we aim to achieve through this summit
- Amplify the patient voice by sharing real clinical trial experiences and fostering open dialogue
- Advance a patient-centered national clinical trial ecosystem that prioritizes patient wellbeing
- Empower patients as active partners by enhancing accessibility, education, and support
- Promote innovations including digital tools, AI, and decentralized models that uplift patient experience
- Ensure ethical, equitable, and compassionate research practices to strengthen trust
- Enhance collaboration among regulators, healthcare providers, academia, industry, and patient communities
- Showcase success stories and lessons learned to inspire progress and highlight patient-centered impact
Key Conference Figures
Expected participation metrics for the summit
50+
Expert Speakers
25+
Participating Countries
40+
Local Entities
30+
International Entities
50+
Scientific Sessions
Featured Speakers
Learn from industry pioneers and thought leaders shaping the future of technology.

Dr. Asma Alfayez
Deputy Chairman, AI Division, KAIMRC
Dr Asma Abdullah Alfayez is a strategic leader in applied artificial intelligence (AI) and innovation. She serves as Deputy Chairman of the Division of AI at King Abdullah International Medical Research Center (KAIMRC). She holds a PhD from University College London (UCL), awarded for a novel research study in AI. Dr Alfayez has published extensively in peer-reviewed journals. She brings over 15 years of experience and professional training within international companies, positioning her as an expert in leading AI divisions, projects, and large-scale initiatives. Her work focuses on translating AI methods into high-impact, real-world applications, including predictive modelling and event forecasting using large-scale, longitudinal data to support evidence-driven decision-making
Dr. Bindhya Cariappa
Executive in Clinical Drug Development & Business Transformation
An executive with expertise in clinical drug development, business growth and transformation within biopharma and healthcare. A global business leader (COO, CSO) who has covered operations in 75 countries, built multi-disciplinary capabilities across Americas, Europe, Middle East, Africa and Asia Pacific. Has been a member of the board of several global entities and is passionate about driving innovation, efficiency, by transforming research through technology.

Dr. Bisher Abuyaseen
VP of Innovation, GreenTech Solutions
A research scientist in experimental medicine with over 20 years of experience ranging between clinical pharmacy practice and basic and clinical research. Dr. Abuyassin obtained his PhD in experimental medicine from the university of British Columbia in Canada in 2018, and since then became an active researcher at KAIMRC with number of additional administrative tasks. He holds currently the position of Biomedical ethics department chairman l and the deputy chairman of the MNGHA-IRB

Dr. Doaa Alsaleh
Physician-Scientist in Clinical Research
A physician-scientist with more than 11 years of experience across academic and industry settings, specializing in clinical research, outcomes research, and clinical trials. She completed a two-year clinical research fellowship at Weill Cornell Medicine in New York, followed by a master’s degree and a PhD in Clinical Research from Mount Sinai, New York. Dr. Alsaleh currently works at the King Abdullah International Medical Research Center (KAIMRC) and has designed and led multiple clinical trials and investigator-initiated studies, in addition to contributing to clinical research training and capacity-building programs in the region.

Dr. Hazem Alshry
Senior Medical Affairs Leader | GCC Medical Leader
A senior medical affairs leader with more than 20 years of extensive experience in the pharmaceutical industry, specializing in Immuno-Oncology, Hematological Malignancies, and rare diseases. Proven expertise in medical and clinical affairs, evidence generation, and stakeholder engagement across GCC and MEA regions, a strong commitment to improving health outcomes and health equity across multiple therapeutic areas, advancing clinical practice change with science-driven and patient focused approaches.

Dr. Jonathan De pierro
Professor of Psychiatry & Associate Director, Center for Stress, Resilience and Personal Growth
Jonathan DePierro, PhD is a Professor of Psychiatry at the Icahn School of Medicine and Associate Director of the Center for Stress, Resilience and Personal Growth at the Mount Sinai Health System in New York, NY. A clinical psychologist by training, Dr. DePierro’s focus is healthcare worker and first responder resilience and mental health. His work has been supported by federal and foundation grants in the United States. Dr. DePierro also serves as Editor in Chief of the journal Psychiatric Quarterly.

Dr. Khalid Alamer
Vice Dean for Research & Innovation | Director of Clinical Trials Unit
The Vice Dean for Research & Innovation at the College of Pharmacy and Director of the Clinical Trials Unit at Imam Abdulrahman Bin Faisal University (IAU), specializing in clinical-trial operations and data management/CTMS infrastructure. He contributes to advancing research capability and supporting multi-sponsor and investigator-initiated clinical trials across multiple institutions.

Prof. Musaad Alshammari
Prof. Musaad Alshammari is a Director of the Research Office at Saudi NIH, where he provides strategic leadership for national health research initiatives. He previously served in academic leadership roles at King Saud University, including Vice Dean of Graduate Studies and Scientific Research. Prof Alshammari is recognized as expert in neuropharmacology, he has authored 60 publications in leading scientific journals. His work spans global collaborations and focuses on advancing research governance, research priorities, and grant management in alignment with Saudi Vision 2030.

Prof. Amro Alhabib
Amro Fayez Al-Habib, MD, FRCSC, MPH, is a Professor of Neurosurgery at King Saud University and a consultant neurosurgeon and spine surgeon in Riyadh, with international training from Harvard University, the University of Toronto, and the University of Calgary, and a clinical and research focus on advanced spine surgery and surgical innovation.

Prof. Ahmed Aljedai

Prof. Ahmed Alaskar
Ahmed Alaskar is a senior hematologist, a university professor, and a clinical researcher with over 20 years of experience. He is a distinguished leader with a significant tenure at the King Abdullah International Medical Research Center (KAIMRC). His leadership has been pivotal in fostering an environment where biomedical innovation thrives. Dr. Alaskar has led teams dedicated to groundbreaking R&D, playing a central role in the development and implementation of biotechnology and shaping-up the clinical trials landscape in Saudi Arabia. Under his guidance, KAIMRC has established national benchmark programs in oncology, stem cell transplantation, and cellular therapy. He was instrumental in launching the Saudi Stem Cell Donor Registry and a national cord blood bank, underscoring his commitment to advancing healthcare and biotechnology. As the President of the Saudi Society of Blood and Marrow Transplantation (SSBMT), he has been at the forefront of shaping the field of hematology in the region. He has cultivated the hematology fellowship training program in Saudi Arabia, demonstrating his dedication to medical education and professional development. Through these contributions, he has guided numerous medical professionals on their journey to excellence while enriching the international medical community with his valuable insights and expertise.

Ms. Jacqueline Johnson North
Jacqueline is CEO of the International Accrediting Organization for Clinical Research (IAOCR), which she co-founded with Dr Martin Robinson in 2011. She has over 20 years of international experience in senior leadership and advisory roles within the biopharmaceutical sector. She brings a strong track record in governance, strategy, and organisational growth, within clinical research, technology, sponsorship, and mining. Jacqueline has completed executive education at Bayes Business School, London, and Kellogg School of Management, Chicago, and is recognised for her focus on innovation, quality, and building high-impact global collaborations.

Mr. Faisal Alrebeishy

Mr. Ahmad Halima
Ahmad Halima is the Director of Clinical Research at PDC CRO in Saudi Arabia, leading clinical operations and strategic delivery across the Middle East and Africa. With over 13 years of experience in clinical research, he has overseen and contributed to more than 100 clinical studies involving over 50,000 patients across a broad spectrum of therapeutic areas, including Oncology, Cardiovascular, CNS Disorders, Immunology, Endocrinology, Psychiatry, Gastroenterology, and Rare Diseases. His expertise spans all phases of clinical development (Phases I–IV), including Real-World Evidence (RWE) studies, with a strong focus on data-driven, patient-centric trial execution. Since joining PDC CRO in 2020, Ahmad has played a pivotal role in scaling regional clinical operations, strengthening sponsor and site partnerships, and expanding operational capabilities across Saudi Arabia and the wider MEA region. He was appointed Director of Clinical Projects in December 2021 and subsequently advanced to Director of Clinical Research in 2022, assuming end-to-end oversight of clinical operations, Phase I–IV programs, and RWE initiatives across multiple countries. Ahmad holds a B.Sc. in Pharmacy & Biotechnology from the German University in Cairo, an Advanced Postgraduate Diploma in Clinical Research & Data Management, and a master's degree in clinical research & data management from the James Lind Institute.

Dr. Yasser Kazzaz

Dr. Sami Aleissa
Spine surgeon with extensive experience in complex spine surgery, currently heading a regional referral center for advanced spine care. Leads comprehensive programs in adult and pediatric spine deformity, spine oncology, and high-risk spinal surgery, with early adoption of navigationassisted, robotic, and awake spine surgery techniques. Led the transformation of the Orthopedic Department at King Abdulaziz Medical City into a high-volume, multi-subspecialty national referral hub, and played a key role in establishing a center of excellence in clinical care, training, and research. Significantly contributed to shaping orthopedic education and training in Saudi Arabia as Chairman of the Orthopedic Scientific Council and Member of the Executive Board of the Saudi Commission for Health Specialties. Led the development and implementation of national training curricula, board reforms, and the accreditation of advanced fellowship programs. Founding President of the Saudi Spine Society, spearheading its establishment and national mission to advance spine education, research, patient safety, and clinical practice guidelines. Organized multiple international spine conferences and numerous scientific events; co-founded the Journal of Spine Practice; and established strategic collaborations with regional and international spine societies. Author of more than 78 peer-reviewed publications and delivered over 100 invited lectures at national and international scientific meetings. Serves as a reviewer for several peer-reviewed journals. Founding member and Executive Board leader of SPINE20, contributing to global advocacy and the development of evidence-based policy recommendations for G20 countries to improve spine care worldwide. Recipient of the King Abdulaziz Medal of Excellence, awarded by royal order of the Custodian of the Two Holy Mosques, King Abdullah bin Abdulaziz Al-Saud.

Dr. Salha Almomen
Dr. Salha Almomen is a board-certified general surgeon and healthcare leader with a strong track record across clinical practice, health policy, and the pharmaceutical industry. Currently serving as External Partnership Director at Pfizer Saudi Arabia, she leads the Pfizer Scientific Institute and drives strategic initiatives focused on, clinical research ecosystem, and medical affairs partnerships. Her work bridges public-private collaboration and contributes to advancing Saudi Arabia’s Vision 2030 goals. Before joining Pfizer, Dr. Almomen held key roles in the Ministry of Health’s Health Technology Assessment Centre, where she shaped national frameworks for evaluating surgical innovations and horizon scanning. She also practiced as a general surgeon and continues to apply her clinical expertise to inform health system improvements. Dr. Almomen holds an MBA in Clinical Leadership from the University of Birmingham and completed her surgical training at King Faisal Specialist Hospital & Research Centre. She is known for her strategic thinking, stakeholder engagement, and deep commitment to improving patient outcomes through sustainable healthcare innovation

Dr. Khalid Alburikan

Dr. Kanan Alshammari
Dr Kanan Alshammari is a consultant of adult medical oncology at King Abdulaziz Medical City – Ministry of National Guard for Health Affairs (MNGHA) in Riyadh, Saudi Arabia, and an Assistant Professor at the College of Medicine of King Saud bin Abdulaziz University (KSAU) for Health Sciences. He is also the director of the NCCN MENA regional coordinating center at MNGHA. He finished his undergraduate training at the University of Otago in New Zealand obtaining his MBChB in 2008. He then obtained the Saudi Board of Internal Medicine in 2014. He did his medical oncology training at McMaster University in Hamilton, Canada, and obtained the Royal College of Physicians and Surgeons of Canada Subspecialty Affiliate certification in 2018. He then did another fellowship in drug development/phase 1 oncology trials and sarcoma in 2019 at the Princess Margaret Cancer Center in Toronto, Canada. He published in the areas of HCC, precision oncology, and has a great interest in sarcoma.

Dr. Jehad Alghamdi (SFDA)
Dr. Jahad Alghamdi is the Executive Director of Benefit and Risk Evaluation at the Saudi Food and Drug Authority (SFDA), where he oversees regulatory assessments and clinical trial approvals. He serves on the National Bioethics Committee and previously led the Saudi Biobank at KAIMRC, playing a pivotal role in advancing precision medicine. A published scientist, Dr. Alghamdi actively contributes to shaping pharmaceutical policy and research ethics.

Dr. Jacki Ying
Jackie Y. Ying was Professor of Chemical Engineering at MIT, and Executive Director of Institute of Bioengineering and Nanotechnology, A*STAR, Singapore. She is Chief Innovation and Research Officer of King Faisal Specialist Hospital & Research Centre, Saudi Arabia. She serves on the Board of Directors of Saudi Arabia’s Research, Development and Innovation Authority, and the Board of Trustees of Princeton University. Prof. Ying has authored over 400 articles (53,500 citations, h index: 109). She is ranked #18 globally in biomedical engineering by ScholarGPS. She has won numerous awards for her research in bioengineering and nanomaterials, including the Mustafa Prize – Top Scientific Achievement Award (2015) and King Faisal Prize in Science (2023). She has been elected to German National Academy of Sciences, Leopoldina, U.S. National Academy of Inventors and U.S. National Academy of Engineering. She is featured on the first Forbes 50 Over 50 Global List of Women Trailblazers (2025).

Dr. Hisham Farouk
Hisham Farouk leads Medical Affairs and Evidence Generation to advance clinical research, innovation, and standards of care across the Kingdom. Earlier, Hisham served as Head of BioPharma at the international level, overseeing medical strategy and operations across four continents: the Middle East, Africa, Asia, and Latin America. In this capacity, he led complex, multi-market portfolios and cross-regional teams, shaping medical strategy across diverse healthcare systems and maturity levels. With more than 23 years of experience in the pharmaceutical industry, Hisham brings deep expertise in medical leadership, real-world evidence, and market-shaping initiatives. He has conceptualized and initiated more than five international studies and led several high-impact real-world evidence programs, including the iCaReMe CVRM registry and iOUTRUN severe asthma study. He has also played a key role in delivering major global and regional studies such as EXACOS, INTERSTELLAR, SABINA, COST OF LUPUS, and the OCS Consensus, alongside multiple practice-shaping clinical consensuses. Hisham has managed over 150 scientific publications and authored more than 20 peer-reviewed manuscripts. He holds an American Board of Specialty Pharmacy (BCPS) certification in Pharmacotherapy and a Master’s degree in Pharmaceutical Sciences (Clinical Pharmacy).

Dr. Farid Askar
Farid's background is Oral surgeon & Medicine, He is Strategic Medical Executive with over 15 years of progressive leadership experience across major pharmaceutical multinationals, With a career spanning from clinical research to executive medical leadership, I have successfully led over 10 Phase II/III clinical trials. I am an expert in navigating the Saudi regulatory and institutional landscape, with significant contributions to national registries and hereditary disorder summits in collaboration with the MOH. I offer a deep understanding of medical governance, talent development, and the strategic alignment of local health needs with global pharmaceutical innovation, I am passionate about utilizing Digital Health solutions and HEOR data to solve complex healthcare challenges and improve patient outcomes in Saudi Arabia.

Dr. Faisal Sanai
Faisal M. Sanai, MD, is a consultant transplant hepatologist at King Abdulaziz Medical City in Jeddah (Saudi Arabia) after completing a fellowship in hepatology and liver transplantation at the Western University in Ontario (Canada) in 2003. He is the editor-in-chief of the Saudi Journal of Gastroenterology and has served on the editorial boards of Liver International and World Journal of Gastroenterology. Dr. Sanai has over 170 publications in both national and international journals as well as over 250 abstracts presented. He has previously served on the board of directors of the Saudi Society for the Study of Liver Diseases and Transplantation (SASLT), the Saudi Gastroenterology Association, and the Liver Disease Research Center at KSU. He is an advisor to the Ministry of Health for the prevention of viral hepatitis, and to the Saudi Health Council for the MASLD National Health Strategy. He has done pioneering work on the formation and publication of national guidelines on various liver diseases. He is the recipient of the annual excellence award by SASLT for the year 2020 for distinguished service in the field of hepatology and liver transplantation.

Dr. Fahad Aljuraibah
1. Associate Dean and Designated Institutional Official (DIO), Postgraduate Education & Academic Affairs at August 2024 till Now Postgraduate Education Deanship, King Saud Bin Abdulaziz University for Health Sciences (KSAU-HS) 2. Director, pediatric residency training program at June 2021 till June 2024 Department of pediatrics, King Abdullah Specialist Children’s Hospital, Riyadh, Ministry of National Guard- Health Affairs

Dr. Emiliano Calvo
Dr. Emiliano Calvo holds a medical degree from the Universidad Autónoma de Madrid (1993) and a PhD from the University of Navarra (2003). He completed his residency in Medical Oncology at the Clínica Universitaria de Navarra (1994–1999) and further specialized in Early Drug Development in Oncology through a Fellowship at the Cancer Therapy and Research Center in San Antonio, Texas (2003–2005). He worked at the Department of Medical Oncology at Vall d’Hebron University Hospital(2005–2008) as Co-Director and Senior Investigator of the Phase I Clinical Trials Unit, and wasthe Program Director for Genitourinary Tumors, Sarcomas, and Brain Tumors. Since 2008, he has served as Director of Clinical Research at the Clara Campal Comprehensive Cancer Center (HM Hospitals), and is also the Founder and Director of the START Madrid group in Spain (Early Phase Clinical Drug Development in Oncology) START now includes four Early Phase Clinical Trial Units: at the Clara CampalComprehensive Cancer Center (Madrid, since 2008), Fundación Jiménez Díaz University Hospital(Madrid, since 2014), HM Delfos Hospital(Barcelona, since 2022) and START Rioja (June 2025).

Dr. Alaa Assem
A strategic and result oriented senior executive with vast and diverse experience in life sciences, having worked in the pharmaceutical, biotech and CRO industries for over 30 years across several leading multinational and regional organization throughout the Middle East and Africa region, including Sandoz, Lilly, Wyeth, Pfizer , Clinart MEA and CTI , in different roles and capacities including: medical affairs, clinical research, regulatory, compliance, government affairs and senior executive roles A strong track record of achievement, with experience in start-ups, building teams, business development and delivery of profitable top and bottom-line growth. Currently, Managing Director of CTI EMEA ( Europe, Middle East and Africa ) , a global full service CRO, Dr Assem brings an extensive and rich experience in clinical research, medical and regulatory affairs, managing clinical trials across the region in accordance with the highest international standards, building clinical research capabilities, expanding geographic footprint and increasing participation of the region in global studies and successfully supporting the introduction of several new innovative products across the ME in different therapeutic areas. His long and diverse industry experience in pharma and CRO is reflected in a strong network of contact with all key stakeholders and understanding of the international and regional business culture and regulatory environment. A physician, holding a MSc of Ophthalmology from Faculty of Medicine – University of Alexandria and an MBA from Maastricht School of Business- Netherlands

Dr. Ahmed Abulaban
Associate Professor and Consultant of Neurology- King Saud bin Abdulaziz University for Health Sciences
Dr. Ahmad Abulaban obtained his bachelor's degree in Medicine and Surgery from King Abdulaziz University, Jeddah, Saudi Arabia. He completed his residency training in Adult Neurology in 2015 at King Abdulaziz Medical City-National Guard-Riyadh. He obtained Multiple Sclerosis and Neuroimmunology Fellowship and Clinical Neurophysiology Fellowship in 2019/2020 from Yale University, USA. Dr. Abulaban is duly certified by the American Board of Clinical Neurophysiology. He was appointed as an Assistant Professor at the College of Medicine, King Saud bin Abdulaziz University for Health Sciences, Riyadh, Saudi Arabia, in 2020. He was appointed as a Consultant Neurologist at King Abdulaziz Medical City, Ministry of National Guard-Health Affairs, Riyadh, Saudi Arabia. He is currently the Residency Training Program Director of Adult Neurology and the Neurosciences and Vision Block Coordinator for Undergraduate Students. Also, he has been serving as the Research Officer of the Saudi Multiple Sclerosis Chapter since 2020. He has multiple publications in the felid of Multiple Sclerosis and Neuroimmunology. He has numerous research awards.

Dr. Abdulrahman Alraizah
Consultant hematologist at King Abdulaziz Medical City, Riyadh
Dr. Abdulrahman Al Raizah is a consultant hematologist at King Abdulaziz Medical City, Riyadh, with a clinical and research focus on thrombosis. He is Program Director of the Adult Hematology Fellowship and Adjunct Assistant Professor at King Saud bin Abdulaziz University for Health Sciences. He completed advanced thrombosis training at the University of Ottawa and holds an MSc in Healthcare Quality and Patient Safety from Harvard Medical School. Dr. Al Raizah serves as site principal investigator for multiple international and multicenter clinical trials in thrombosis and related hematologic disorders. He chairs the institutional Thrombosis Program and leads thrombosis-focused quality improvement and guideline implementation initiatives, including the use of artificial intelligence to improve thrombosis care.

Prof. Khalid Eljaaly
Associate Professor & Chairman, Department of Pharmacy Practice
Prof. Khalid Eljaaly, PharmD, MS, BCPS, BCIDP, FCCP, FIDSA is an associate professor and chairman of Department of Pharmacy Practice at King Abdulaziz University. He is a consultant infectious disease pharmacist and R3 pharmacy residency in infectious disease program director at King Abdulaziz University Hospital. He completed PGY2 infectious disease pharmacy residency at BIDMC, a Harvard Medical School-affiliated hospital, and both antibiotic stewardship pharmacy fellowship and Master of Science in clinical translational research at University of Arizona.

Prof. Mohammed Alshahrani
Professor of Emergency & Critical Care Medicine | Dean, College of Medicine

Dr. Riad El Fakih
Consultant Hematologist & Chair of Clinical Research Department
Riad El Fakih, MD, is a consultant in the Department of Hematology, Stem Cell Transplant & Cellular Therapy at the Oncology Center of King Faisal Specialist Hospital and Research Center, Riyadh. He is also the Chair of Clinical Research Department at KFSH&RC. Dr. El Fakih is American Board certified in Internal Medicine, Hematology and Oncology. He has special interest in cellular therapies and lymphoid neoplasms. Dr. El Fakih has authored more than 130 peer-reviewed publications, a handbook of Bone Marrow Transplantation, and 6 Hematology book chapters. He contributes to vital hospital committees and NCCN guidelines, all while serving as an Associate Editor and reviewer for prestigious journals, cementing his commitment to advance medical excellence and patient care.

Dr. Suad Hannawi
Consultant Rheumatologist & Chair of Rheumatology
Suad Hannawi (BSC, MD, MRCP (UK), FRCP (UK), MIPH, and PhD); a Consultant Rheumatologist and Chair of Rheumatology at Emirates Health Services (EHS), UAE, and President of the GCC Association of Immunology and Rheumatology (GCC-AIR), Chair of the EHS and Dubai Research Ethics Committees (REC) and Deputy Chair of the Central REC at the Ministry of Health and Prevention (MOHAP).

Ms. Heather Rice
Director of Research and Innovation
Graduate of NHS national management training scheme with diverse experience across health services and local government. LLB and MA in Health Services Management. Recipient of prestigious fellowships including King's Fund Leadership Programme, Nuffield Trust Fellowship, and International Women's Forum Fellowship. Current role within Rotherham, Doncaster and South Humber NHS Trust for 11+ years, specializing in clinical and political leadership support.

Prof. Mazen Hassanain
Vice President for Research & Innovations | Founder & Managing Director, SaudiVax
Professor Mazen Hassanain is the Vice President for Research and Innovations at Fakeeh Care Group, a large private healthcare provider with comprehensive services and a leader of academic medicine in the private sector. He is also the founder and Managing Director of SaudiVax, a pioneer Saudi biotechnology company responsible for developing and manufacturing high-quality and affordable vaccines & biopharmaceuticals. He is a Professor of Surgery and a hepatobiliary and solid organ transplant surgery Consultant, an Adjunct Professor at the Department of Pediatrics at Baylor College of Medicine, and a formal Adjunct Professor at the Department of Oncology at McGill University. He is an experienced clinician-scientist with a demonstrated history of working in hospitals, research centers, and the healthcare industry. Skilled in collaborative translational, immunology, and oncology research and development projects and is success-driven. Strong educational background with a Doctor of Philosophy (Ph.D.) focused on moving scientific research from bench to bed. He received his medical degree with honours from King Saud University and did his surgical residency and transplantation fellowship at McGill University, Montreal, Canada. He is a Fellow of The Royal College of Physicians of Canada, a Fellow of the American College of Surgeons, a member of the American Society of Transplant Surgery Associations, and the American Association for Cancer Research. Before SaudiVax, Prof. Hassanain worked at the Saudi Ministry of Health from 2012 to 2014 as the Director-General of the General Administration of Research and Studies. He led the health research strategy for Saudi Arabia and initiated the health services research center, the central ethics review board, and the national biobank. In the last 15 years, he has presented 60 oral presentations/posters at national and international meetings, resulting in 120 peer-reviewed publications. He has also supervised hundreds of graduate and postgraduate students in Saudi Arabia and Canada.

Prof. Reem Alkhalifah
Professor of Pediatrics & Pediatric Endocrinology
Prof. Reem Abdullah H. Al Khalifah is a Professor of Pediatrics and Pediatric Endocrinology at King Saud University, Riyadh. She completed subspecialty fellowships in endocrinology, metabolism, and pediatric obesity at McGill University, and McMaster University, where she also earned a Master of Science in Health Research Methodology (Clinical Epidemiology). Her research focuses on pediatric diabetes, obesity, endocrine disorders, and clinical epidemiology, with more than 60 peer-reviewed publications in high-impact journals. She has received multiple national and international awards, serves as a peer reviewer and academic editor, and is an active member of leading professional societies including the Endocrine Society, ISPAD, and the Saudi Society of Endocrinology and Metabolism. Currently, she is the head of the clinical trials section at King Saud University Medical City.

Prof. Waleed Alhazzani
CEO, Scientific Research Center & Professor of Critical Care Medicine
Prof. Waleed Alhazzani is CEO of the Scientific Research Center at the Ministry of Defense Health Services in Saudi Arabia and Professor of Critical Care Medicine. He has published more than 250 peer-reviewed papers. He has led the development of international and national clinical practice guidelines with leading critical care societies worldwide, including SCCM, ESICM, ATS, and the Surviving Sepsis Campaign. Prof. Alhazzani is a CIHR, SNIH, and RDIA–funded clinical trialist and has received multiple awards for research and teaching, including several Presidential Citations from SCCM and the Clive Kearon Research Award. His work continues to shape critical care practice and advance research systems both in Saudi Arabia and globally.
Conference Activities
Engaging sessions and interactive opportunities
Main Sessions
Scientific sessions led by a select group of local and international experts, presenting strategic visions and future trends in clinical trials and medical research.
Discussion Panels
Include interactive sessions bringing together decision-makers, researchers, and practitioners to discuss practical challenges and best practices, and exchange experiences across different sectors.
Scientific Presentations
Activities dedicated to presenting recent research and scientific studies, allowing researchers to share their results, discuss research methodologies, and enhance scientific communication and build research collaboration opportunities.
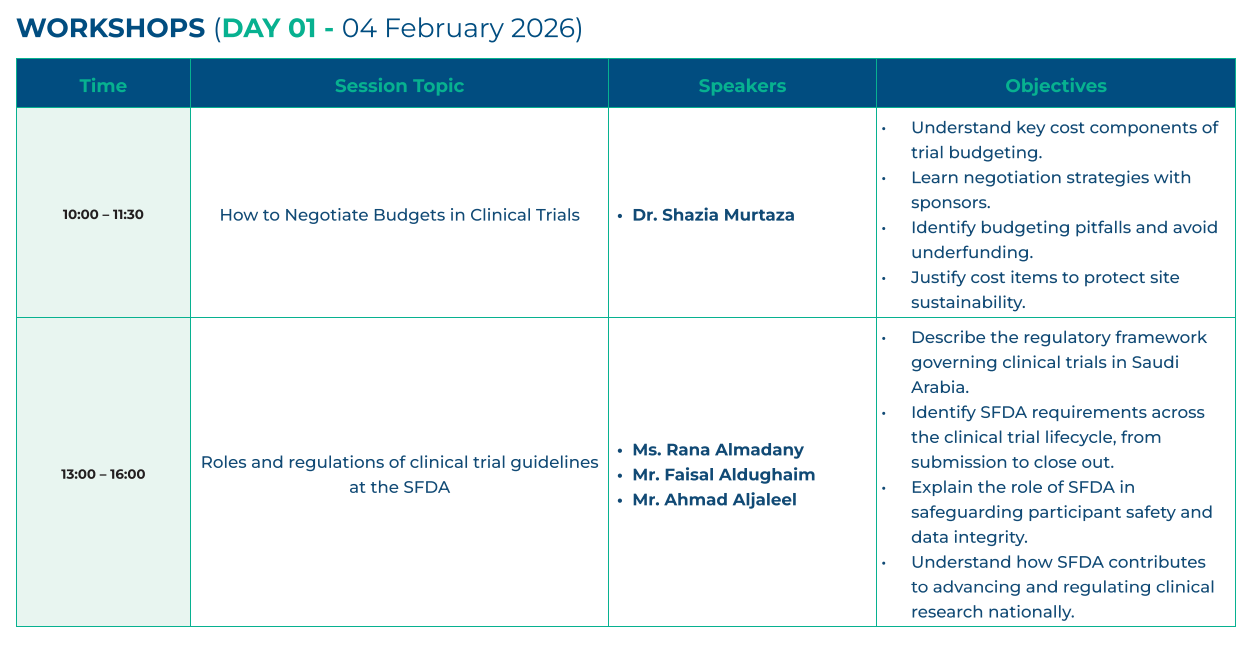
Scientific program of the conference
Two days of insightful sessions, workshops, and networking opportunities.






Our Sponsors
Platinum Sponsors
Gold Sponsors


Silver Sponsors



Exhibition Sponsors






Register for The 3rd International Conference on Clinical Trials 2026
Secure your spot at The 3rd International Conference on Clinical Trials of the year.
